 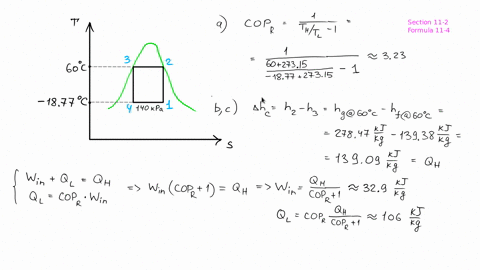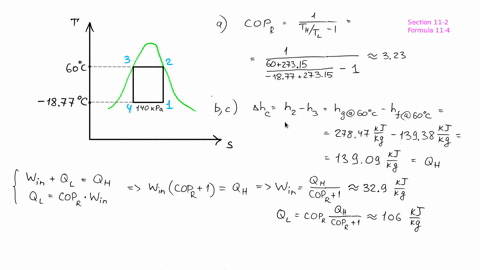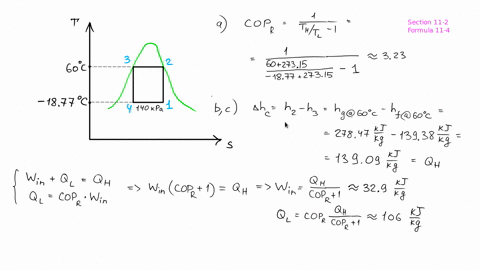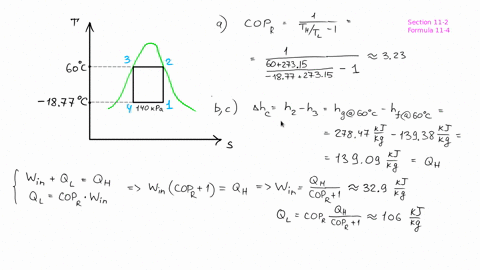   
The piston now slowly expands, maintaining constant temperature to a final pressure of 400 kPa in a reversible process. # -*- coding: utf8 -*- from _future_ import division #Example: 8.2 ''' A cylinder/piston setup contains 1 L of saturated liquid refrigerant R-410a at 20◦C. Find the pressure after compression, before the heat rejection process, and determine the COP for the cycle.''' #Variable Declaration: Th = 60 #temperature at which heat is rejected from R-134a Tl = 0 #temperature at which heat is absorbed into the R-134a s1 = 1.7262 #specific entropy at 0 Celsius s3 = 1.2857 #specific entropy at 60 celsius s4 = s3 #process of state change from 3-4 is isentropic #Calculations: s2 = s1 #process of state change from 1-2 is isentropic P2 = 1400 + ( 1600 - 1400 ) * ( 1.7262 - 1.736 ) / ( 1.7135 - 1.736 ) #pressure after compression in kPa B = ( Th + 273 ) / ( Th - Tl ) #coefficient of performance of refrigerator #Results: print "Pressure after compression is P2: ", round ( P2, 1 ), 'KPa' print "Coefficient of performance of refrigerator: ", round ( B, 2 ) The heat is rejected from the R-134a at 60◦C and ends up as saturated liquid. Heat is absorbed into the R-134a at 0◦C, during which process it changes from a two-phase state to saturated vapor. # -*- coding: utf8 -*- from _future_ import division #Example: 8.1 ''' Consider a Carnot-cycle heat pump with R-134a as the working fluid. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
